SIGMA-GBS™ – Group B Strep
320,000 babies develop invasive Strep B infection. 90,000 babies under 3 months die from Strep B infection. (GBSS.org.uk). Group B Strep infection is preventable and should be detected sooner before the baby is born.
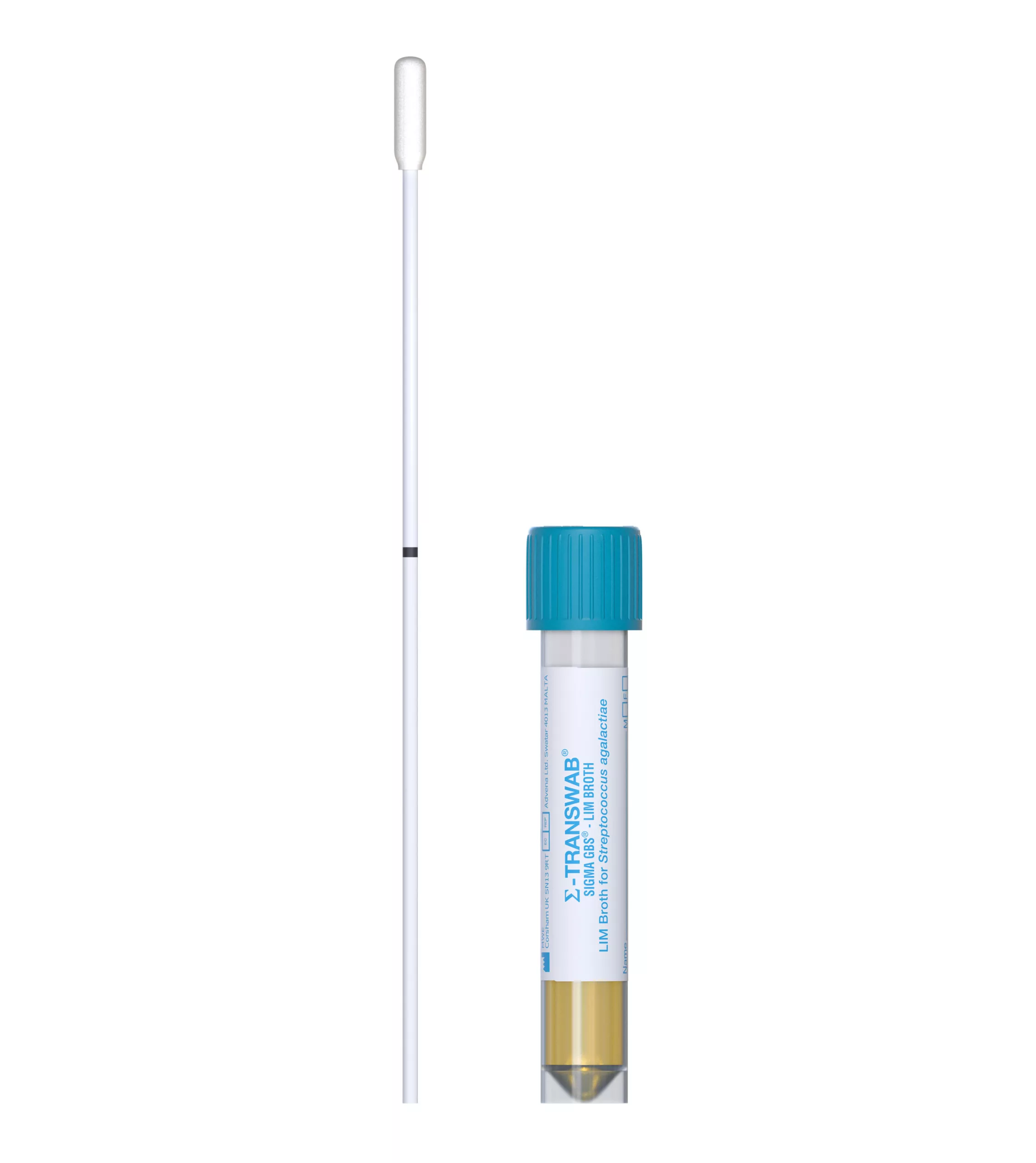
SIGMA GBS™ is a swab-based device. It enables direct collection and rapid processing of specimens for screening for Streptocccus agalactiae. It has an enrichment broth, which means that after incubation, the specimen is ready for direct plating on chromogenic agar for the detection and identification of Streptocccus agalactiae, or for testing by molecular techniques such as PCR.
Sigma-GBS™ can be stored at ambient temperature allowing the products to be kept near the patients ready for testing. It includes a 2ml vial of Lim broth (for the enrichment of Group B Streptococcus) and a swab which can be snapped into the vial.
Screening for Streptocccus agalactiae is normally performed in late pregnancy. The swab is used to collect specimen(s) in normal way from vagina and/or rectum.
- Lim Broth Medium
- Enrichment of specimens ready for direct plating on chromogenic agar
- Interfering bacteria inhibited, including E. coli
- Liquid format compatible with automated processing platforms
- Simpler processing and faster turnaround
- Convenient Snap ‘n’ Cap format for easy specimen handling
- Convenient format for screening for Group B Streptococcus colonisation
| Product Code | Description | Cap Colour | Swab Length | Pack Size | Expiry (Months) |
|---|---|---|---|---|---|
| MWGBS | SIGMA GBS™ - Standard Plastic Foam Printed Breakpoint - 12x80 Vial - 2ml LIM Broth | Turquoise | 15cm | 125 | 9 |
| MWGBST | SIGMA GBS™ - 12x80 Labelled Vial - 2ml LIM Broth | Turquoise | n/a | 50 | 9 |
Contact us
Phone
Address
Corsham, Wiltshire
England
SN13 9RT
Have a question or would like to find out about one of our products? Please fill in the form below and we’ll be in touch soon.
Product enquiry
For buying enquiries, or for more information about this product, please fill in the form below and we’ll be in touch soon.
Let’s see...
We can direct you to the best place. Would you like help with: