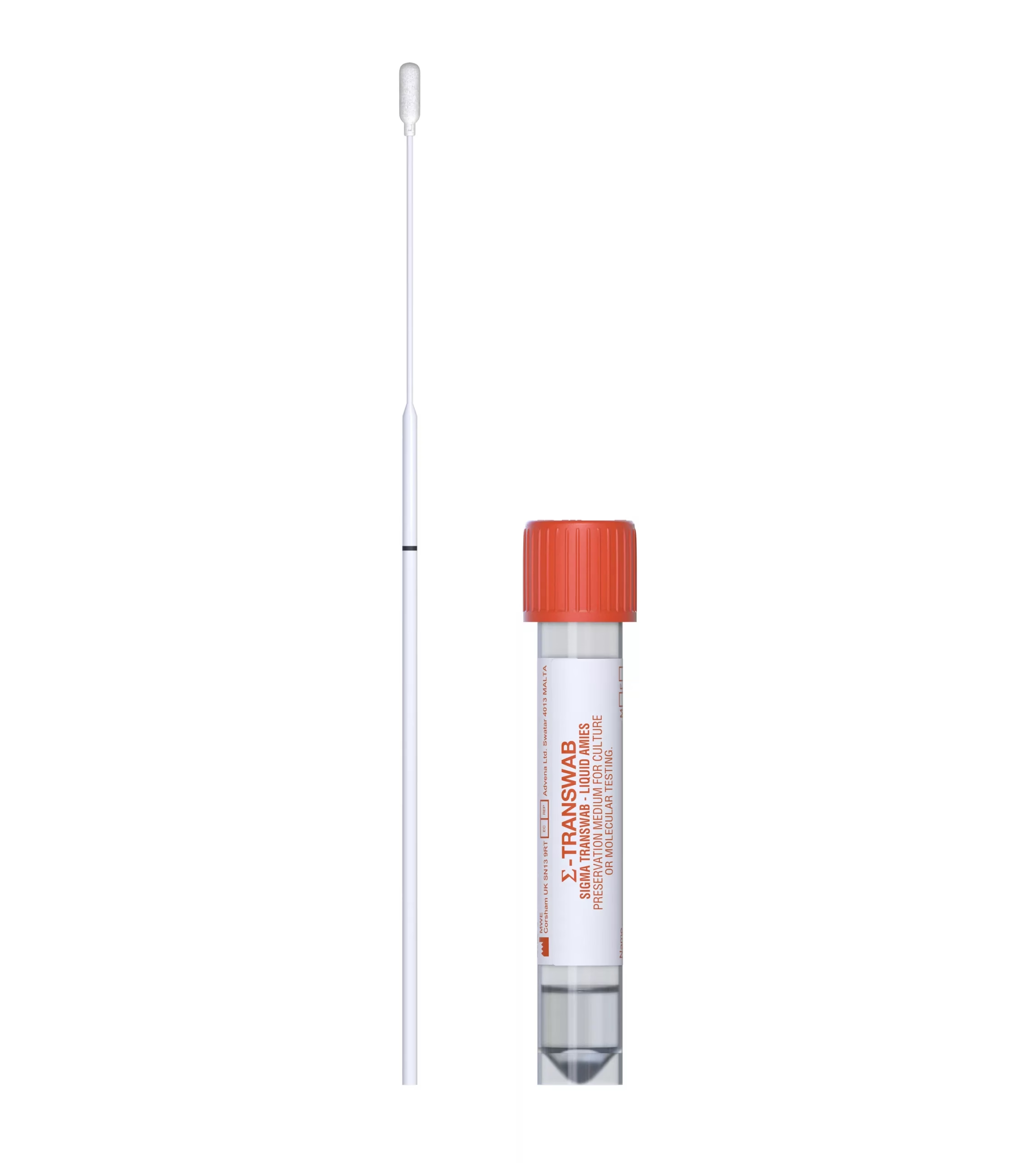
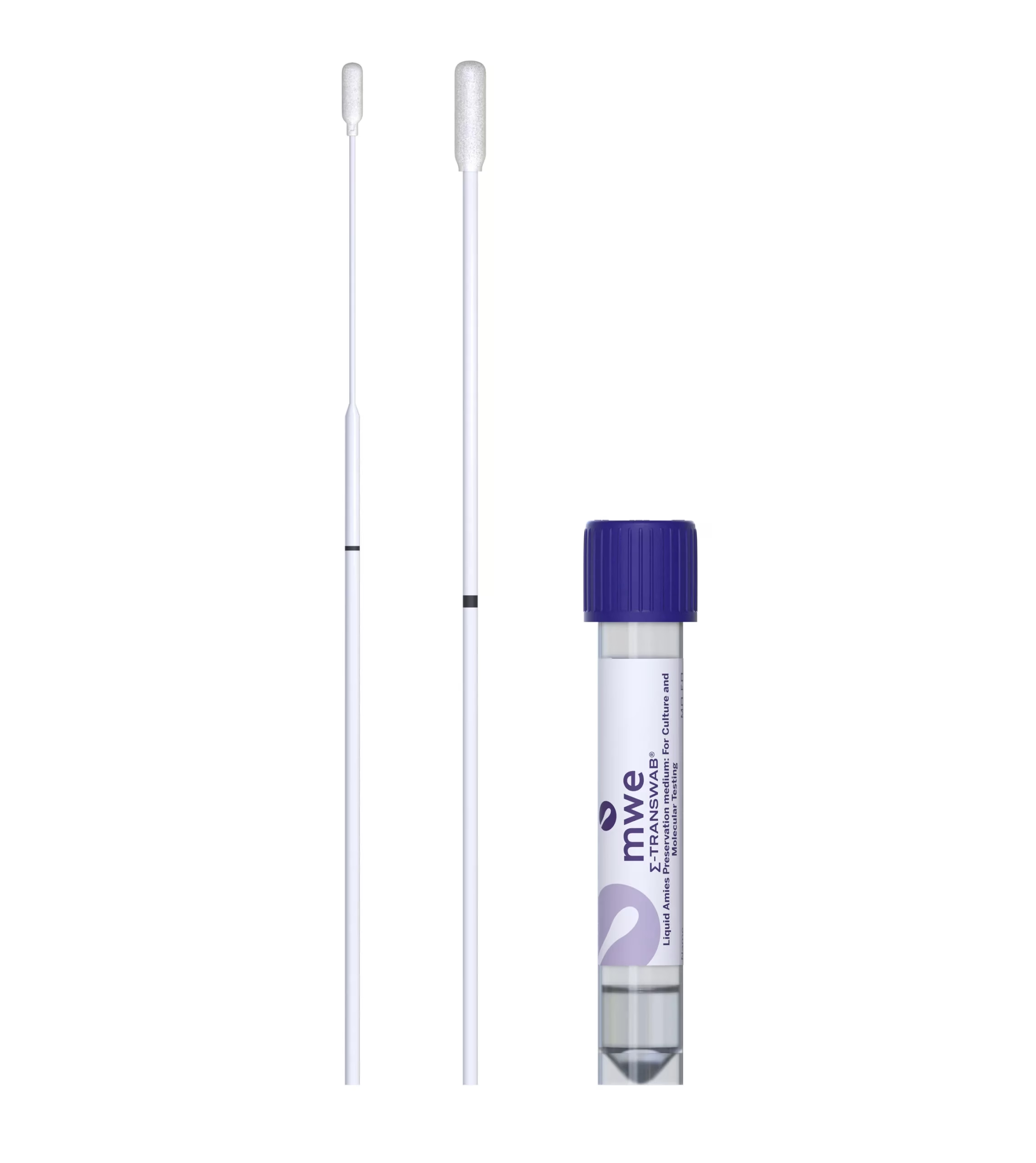
SIGMA-TRANSWAB®
SIGMA TRANSWAB® for bacterial & viral specimen collection and transport used in a clinical setting for wounds, skin, throat and other swabbing with MRSA and ENT/STD options. SIGMA-TRANSWAB® is the world’s leading liquid amies transport for automated and conventional processing.
Many laboratories are moving from gel to liquid as the liquid allows complete dispersion of the sample. Uniform suspension can be aliquoted for multiple tests and specimens can be pooled. Ideal for epidemiology and surveillance studies
M40-A2 compliant (CLSI standard M40-A2: Quality Control of Microbiological Transport Systems Transport Devices).
The specimen is collected by swab & snapped into the tube of liquid amies transport medium. This process is enhanced by the incorporation of an open-celled foam-tipped swab which allows complete flow through of the liquid medium, reagents and microorganism. This increases the sensitivity of any diagnostic procedures.
The microorganisms from the initial specimen are released from the swab & dispersed throughout the medium, producing a uniform suspension ready for use, whether it’s for direct inoculation onto plates or into a broth, or for use with the many molecular diagnostics currently available.
The Original
TRANSWAB® was the first commercially produced gel transport swab, and for many years has led the field for reliable microbiological specimen collection and transport. A programme of continuous development has kept TRANSWAB® ahead of changes in laboratory science and regulatory requirements, so that it remains a trusted partner in the diagnosis of infectious diseases.
Liquid amies media in a shatter proof vial.
Recommended Applications:
Microbiological specimen collection and transport used in a clinical setting for wounds, skin, throat and other swabbing with MRSA and ENT/STD options.
Clearances:
FDA 510(k)
- The MWE open-celled flow through foam bud allows full access and release of sampled organisms for skin and wounds, improving results with no requirement to moisten the swab before taking the sample.
- 1ml, 2ml & 3ml vials compatible with conventional and automated processing.
- The 2ml & 3ml allows additional testing from the same patient sample if required
- Cap colours (Purple White and Orange) allow easy identification for point of care sampling and in the lab:
Purple: Universal Testing
White: MRSA
Orange: ENT & STD - Cap incorporates an ingenious swab capture mechanism which enables automatic removal of the swab for diagnostic testing.
-
| Product Code | Description | Cap Colour | Swab Length | Pack Size | Expiry (Months) |
|---|---|---|---|---|---|
| MRSA | |||||
| MW167S | SIGMA TRANSWAB® - 2 Standard Plastic Foam Printed Breakpoint - 12x80 Vial -1.5ml Amies Liquid Medium | White | 15cm | 125 | 24 |
| MW176S3 | SIGMA TRANSWAB® - 1 Standard Plastic Foam Printed Breakpoint + 2 Red Plastic Foam - 12x80 Vial - 1.5ml Liquid Amies Medium | White | 15cm | 125 | 24 |
| MW176S | SIGMA TRANSWAB® - Standard Plastic Foam Printed Breakpoint - 12x80 Vial - 1ml Liquid Amies Medium | Purple | 15cm | 125 | 24 |
| MW176PF | SIGMA TRANSWAB® - Standard Flockswab Printed Breakpoint - 12x80 Vial - 1ml Liquid Amies Medium | Purple | 15cm | 125 | 24 |
| MW176S2ML | SIGMA TRANSWAB® - Standard Plastic Foam Printed Breakpoint - 12x80 Tube - 2ml Liquid Amies Medium | Purple | 15cm | 125 | 24 |
| MW176SE2 | SIGMA TRANSWAB® - 1 Standard Foam+ 1 ENT Foam - 12x80 Vial - 1.5ml Liquid Amies Medium | Purple | 15cm | 125 | 24 |
| MW176T3 | SIGMA TRANSTUBE® - 12x80 Vial - 3ml Liquid Amies Medium | Purple | N/A | 200 | 24 |
| MW178PF | SIGMA TRANSWAB® - Ultrafine Flexible Minitip Flockswab Printed Breakpoint - 12x80 Vial -1ml Liquid Amies Medium | Light Blue | 15cm | 125 | 24 |
| ENT (Ears, Nose, Throat) and STD | |||||
| MW177S | SIGMA TRANSWAB® - ENT Minitip Foam Printed Breakpoint - 12x80 Vial - 1ml Liquid Amies Medium | Orange | 15cm | 125 | 24 |
| MW177PF | SIGMA TRANSWAB® - ENT Minitip Flockswab Printed Breakpoint - 12x80 Vial - 1ml Liquid Amies Medium | Orange | 15cm | 125 | 24 |
| MW177S2ML | SIGMA TRANSWAB® - ENT Minitip Foam Printed Breakpoint - 12x80 Vial - 2ml Liquid Amies Medium | Orange | 15cm | 125 | 24 |
| MW176M | SIGMA TRANSTUBE® - 12x80 Vial - 1ml Liquid Amies | Purple | N/A | 50 | 24 |
| MW178PF | SIGMA TRANSWAB® - Ultrafine Flexible Minitip Flockswab Printed Breakpoint - 12x80 Vial -1ml Liquid Amies Medium | Light Blue | 15cm | 125 | 24 |
Contact us
Phone
Address
Corsham, Wiltshire
England
SN13 9RT
Have a question or would like to find out about one of our products? Please fill in the form below and we’ll be in touch soon.
Product enquiry
For buying enquiries, or for more information about this product, please fill in the form below and we’ll be in touch soon.
Stay informed about industry news, current events, and MWE’s industry leading product ranges that enable the most accurate and reliable results.
MWE requires your contact information in order to send you the requested content and to contact you about our services. You may unsubscribe from these communications at any time. For information on how we use your data, please see our Privacy Policy.
Let’s see...
We can direct you to the best place. Would you like help with: